Lectures in THERMOCHEMISTRY
 Lecture 1: Definition
Lecture 1: Definition Lecture 2: Exothermic And Endothermic Reactions
Lecture 2: Exothermic And Endothermic Reactions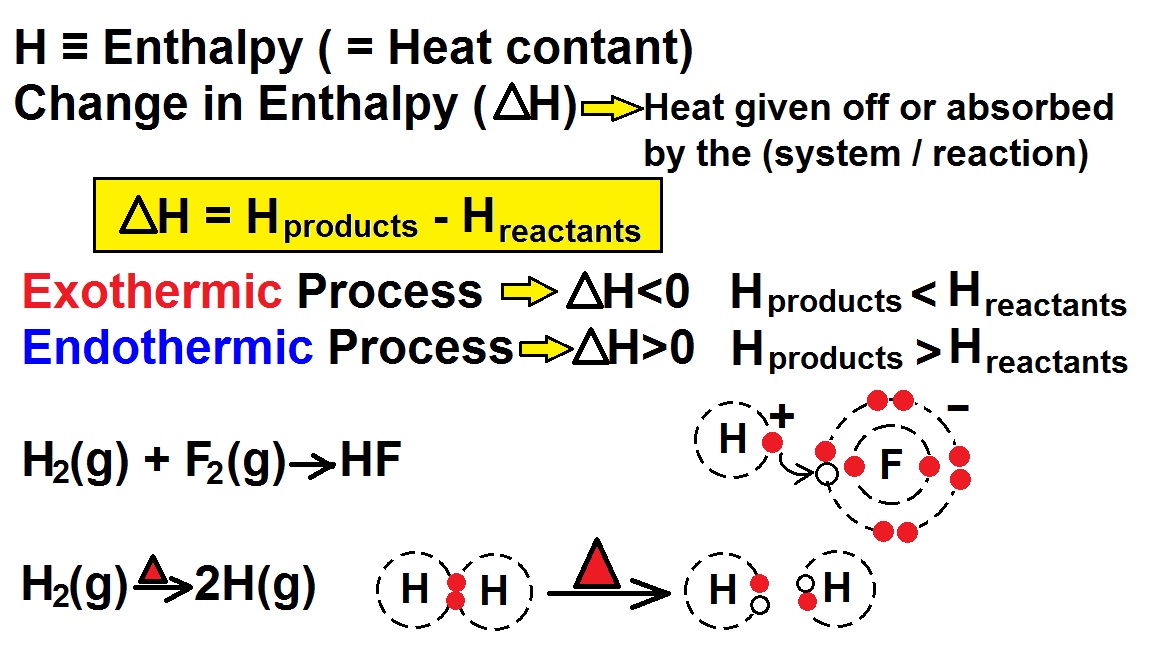 Lecture 3: Enthalpy: Definition
Lecture 3: Enthalpy: Definition Lecture 4: Enthalpy: Physical And Chemical Change
Lecture 4: Enthalpy: Physical And Chemical Change Lecture 5: Enthalpy: Example 1 (Combustion Of Methane)
Lecture 5: Enthalpy: Example 1 (Combustion Of Methane) Lecture 6: Enthalpy: Example 2 (Combustion Of Ethane)
Lecture 6: Enthalpy: Example 2 (Combustion Of Ethane) Lecture 7: Enthalpy: Example 3 (Combustion Of Propane)
Lecture 7: Enthalpy: Example 3 (Combustion Of Propane) Lecture 8: Enthalpy: Hess' Law
Lecture 8: Enthalpy: Hess' Law Lecture 9: Enthalpy: Hess' Law: Example 1A
Lecture 9: Enthalpy: Hess' Law: Example 1A Lecture 10: Enthalpy: Hess' Law: Example 1B
Lecture 10: Enthalpy: Hess' Law: Example 1B Lecture 11: Enthalpy: Hess' Law: Example 2
Lecture 11: Enthalpy: Hess' Law: Example 2 Lecture 12: Enthalpy: Hess' Law: Example 3
Lecture 12: Enthalpy: Hess' Law: Example 3 Lecture 13: Enthalpy: Hess' Law: Example 4
Lecture 13: Enthalpy: Hess' Law: Example 4 Lecture 14: Heat Released (Evolved) In A Reaction 1
Lecture 14: Heat Released (Evolved) In A Reaction 1 Lecture 15: Heat Released (Evolved) In A Reaction 2
Lecture 15: Heat Released (Evolved) In A Reaction 2 Lecture 16: Heat Released (Evolved) In A Reaction 3
Lecture 16: Heat Released (Evolved) In A Reaction 3 Lecture 17: Heat Released (Evolved) In A Reaction 4
Lecture 17: Heat Released (Evolved) In A Reaction 4 Lecture 18: Heat Released (Evolved) In A Reaction 5
Lecture 18: Heat Released (Evolved) In A Reaction 5 Lecture 19: Heat Released (Evolved) In A Reaction 6
Lecture 19: Heat Released (Evolved) In A Reaction 6 Lecture 20: Heat Released (Evolved) In A Reaction 7
Lecture 20: Heat Released (Evolved) In A Reaction 7 Lecture 21: Heat And Calorimetry: Basic Definition
Lecture 21: Heat And Calorimetry: Basic Definition Lecture 22: Heat And Calorimetry: Example 1
Lecture 22: Heat And Calorimetry: Example 1 Lecture 23: Heat And Calorimetry: Example 2
Lecture 23: Heat And Calorimetry: Example 2 Lecture 24: Heat And Calorimetry: Example
Lecture 24: Heat And Calorimetry: Example Lecture 25: The Bomb Calorimeter (Constant Volume)
Lecture 25: The Bomb Calorimeter (Constant Volume) Lecture 26: Constant Pressure Calorimeter
Lecture 26: Constant Pressure Calorimeter Lecture 27: Combustion In A Bomb Calorimeter: Ex. 1
Lecture 27: Combustion In A Bomb Calorimeter: Ex. 1 Lecture 28: Combustion In A Bomb Calorimeter: Ex. 2
Lecture 28: Combustion In A Bomb Calorimeter: Ex. 2 Lecture 29: Combustion In A Bomb Calorimeter: Ex. 3
Lecture 29: Combustion In A Bomb Calorimeter: Ex. 3 Lecture 30: Constant Pressure Calorimeter: Ex. 1
Lecture 30: Constant Pressure Calorimeter: Ex. 1 Lecture 31: Constant Pressure Calorimeter: Ex. 2
Lecture 31: Constant Pressure Calorimeter: Ex. 2 Lecture 32: Constant Volume Calorimeter (Bomb Calorimeter)
Lecture 32: Constant Volume Calorimeter (Bomb Calorimeter) Lecture 33: Heat Of Solution (Enthapy Of Solution)
Lecture 33: Heat Of Solution (Enthapy Of Solution) Lecture 34: Heat Of Dilution
Lecture 34: Heat Of Dilution Lecture 35: Heat Of Dilution: Danger!
Lecture 35: Heat Of Dilution: Danger! Lecture 36: The Cold Pack
Lecture 36: The Cold Pack Lecture 37: The Hot Pack
Lecture 37: The Hot Pack


